The most severe FDA penalty for non-compliance just might come in 2013.
It's called a "NO SALE" order, and it's reserved for tobacco-selling businesses with "repeat and serious violations."
You might think there's no need to worry about your store, but let's consider the process that's underway.
FDA's enforcement system is no longer in "start-up" mode. It has completed over 131,000 compliance checks (95,000 in 2012). And we'll certainly see even more checks in 2013 now that enforcement systems are active in 39 states plus the District of Columbia.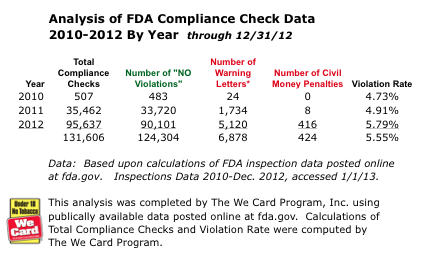
The severity of FDA's penalty structure is based on two things:
1. number of violations; and,
2. the time period in which violations occur
(such as within 24 months, 36 months or 48 months.)
 Get We Card's Guide to Best Practices -- an interactive PDF. To obtain the Guide, please complete our survey first.
Get We Card's Guide to Best Practices -- an interactive PDF. To obtain the Guide, please complete our survey first.
Take We Card Online Training -- an Employee Training Course is available with FDA content. Manager Training Course and a Refresher Training Course are available now.
ID Check-Up -- a confidential mystery shopping service to check your store's performance at carding customers.
We Card's Get Your Store Ready for FDA Enforcement
We Card's Federal Law and FDA Regulations summary
State Law Summaries Find out about the laws in your state -- including fines and penalties.
State Sign Requirement -- In some states, retailers are fined for not having the legally required signs posted. Find out what the sign looks like in your state.
Minor License Visual Characteristics -- Find out what the minor license in your state looks like.
Company Tobacco Sales Policy and Employee Acknowledgement of Company Policy -- We Card's recommended template you can use to customize for your company.
Warning Letter
If a tobacco retailer receives an FDA Warning Letter (FDA observed violations during a store visit), get ready for a 2nd FDA inspection. They have followed through on the promise to re-visit stores observed to be in violation.
Follow Up Compliance Visits
On the second inspection, if a retailer fails and gets cited for not checking ID, selling to a minor or other violations (advertising or merchandising), the violations add up. Why? It appears FDA counts any violations associated with the Warning Letter as "one violation." For all future store inspections, each violation observed is counted separately. With most "failed" inspections showing at least two violations, and some showing four, this second inspection can bring violation 3, 4 or 5 very quickly.
See the FDA Fines & Penalties chart shown at top right.
Fail the second inspection, which triggers another repeat store inspection, and we're already climbing to the most severe penalty level: $10,000 or perhaps a "No Sale Order."
The point is that it really isn't difficult to rise up the penalty chart quickly if your store is a repeat violator.
Right now, several retailers have three or more violations within a 24-month period; a few $5,000 fines have been issued, including a whopping $10,000 fine. Another inspection or two and poor compliance will lead to the "NO SALE" penalty threshold.
With the average FDA Civil Money Penalty at approximately $639, it's probably safe to say that this average will rise . . . particularly now that the number of inspections and re-inspections will steadily increase in the coming year.
I suppose you can guess where I'm headed with this preview: Penalties are avoidable with compliance. Compliance happens with education. Educating all of your employees on the right way to handle age-restricted products, especially FDA regulated products, through proper and effective training is small measure compared to the risks of being on the list of retailers slated for re-inspection.
Doug Anderson
President
The We Card Program, Inc.
© 2013 Copyright - The We Card Program, Inc. All Rights Reserved.